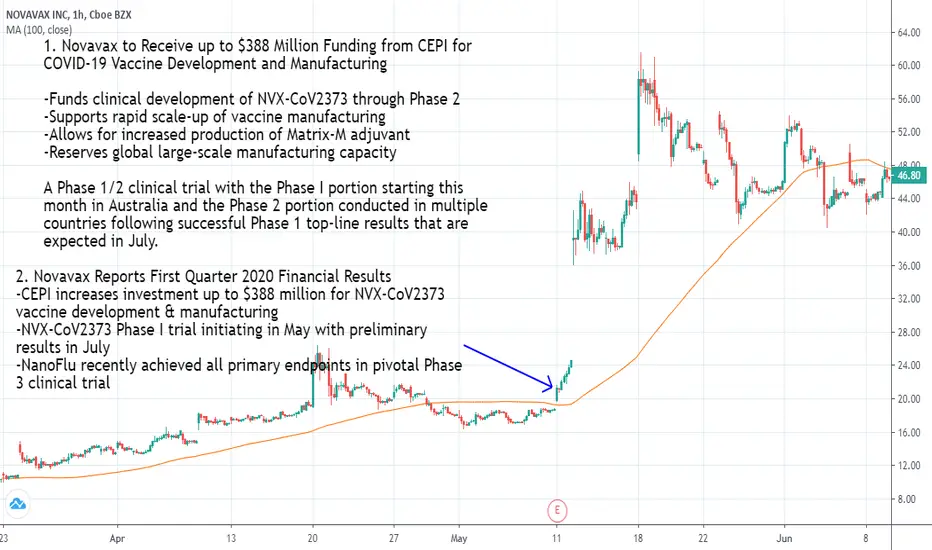
1. Novavax to Receive up to $388 Million Funding from CEPI for COVID-19 Vaccine Development and Manufacturing
-Funds clinical development of NVX-CoV2373 through Phase 2
-Supports rapid scale-up of vaccine manufacturing
-Allows for increased production of Matrix-M adjuvant
-Reserves global large-scale manufacturing capacity
A Phase 1/2 clinical trial with the Phase I portion starting this month in Australia and the Phase 2 portion conducted in multiple countries following successful Phase 1 top-line results that are expected in July.
ir.novavax.com/news-releases/news-release-details/novavax-receive-388-million-funding-cepi-covid-19-vaccine
2. Novavax Reports First Quarter 2020 Financial Results
-CEPI increases investment up to $388 million for NVX‑CoV2373 vaccine development & manufacturing
-NVX‑CoV2373 Phase I trial initiating in May with preliminary results in July
-NanoFlu recently achieved all primary endpoints in pivotal Phase 3 clinical trial
ir.novavax.com/news-releases/news-release-details/novavax-reports-first-quarter-2020-financial-results
-Funds clinical development of NVX-CoV2373 through Phase 2
-Supports rapid scale-up of vaccine manufacturing
-Allows for increased production of Matrix-M adjuvant
-Reserves global large-scale manufacturing capacity
A Phase 1/2 clinical trial with the Phase I portion starting this month in Australia and the Phase 2 portion conducted in multiple countries following successful Phase 1 top-line results that are expected in July.
ir.novavax.com/news-releases/news-release-details/novavax-receive-388-million-funding-cepi-covid-19-vaccine
2. Novavax Reports First Quarter 2020 Financial Results
-CEPI increases investment up to $388 million for NVX‑CoV2373 vaccine development & manufacturing
-NVX‑CoV2373 Phase I trial initiating in May with preliminary results in July
-NanoFlu recently achieved all primary endpoints in pivotal Phase 3 clinical trial
ir.novavax.com/news-releases/news-release-details/novavax-reports-first-quarter-2020-financial-results
Penafian
Maklumat dan penerbitan adalah tidak dimaksudkan untuk menjadi, dan tidak membentuk, nasihat untuk kewangan, pelaburan, perdagangan dan jenis-jenis lain atau cadangan yang dibekalkan atau disahkan oleh TradingView. Baca dengan lebih lanjut di Terma Penggunaan.
Penafian
Maklumat dan penerbitan adalah tidak dimaksudkan untuk menjadi, dan tidak membentuk, nasihat untuk kewangan, pelaburan, perdagangan dan jenis-jenis lain atau cadangan yang dibekalkan atau disahkan oleh TradingView. Baca dengan lebih lanjut di Terma Penggunaan.