Panjang
$SIOX PT 10-19 and higher
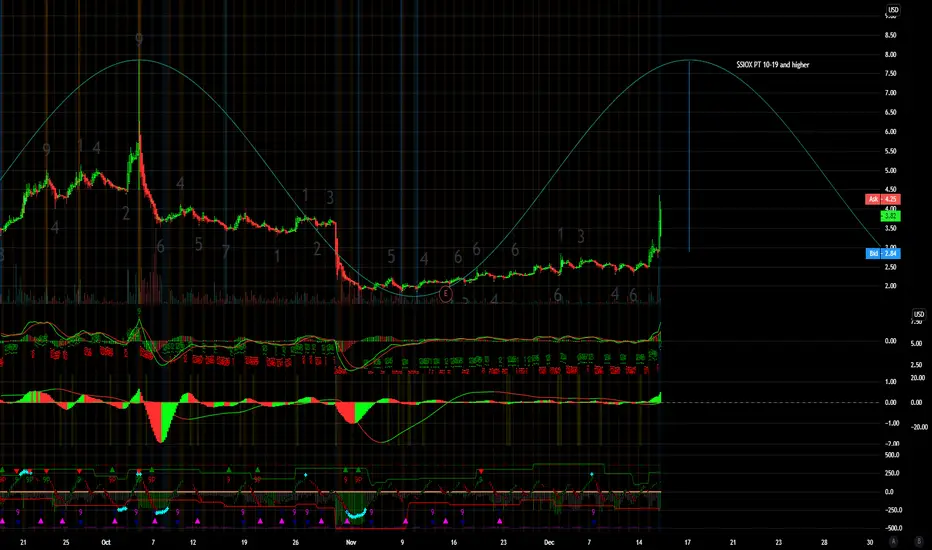
Sio Gene Therapies Announces Positive Six-Month Follow-Up Data from Low-Dose Cohort of Phase 1/2 Trial of AXO-AAV-GM1 for GM1 Gangliosidosis
- Generally well-tolerated with a favorable safety profile in five patients
- Serum beta-galactosidase enzyme activity increased in all patients at all timepoints between Day 7 and Month 6, representing an approximate doubling in enzyme activity after gene transfer
- At Month 6, enzyme activity restored to 23-57% (mean: 38%) of normal reference levels
- All five children demonstrated signs of clinical disease stability as assessed by Vineland-3 Growth Scale Value, Upright and Floor Mobility, and Clinical Global Impression (CGI) scales
- High-dose cohort initiated in November 2020; two patients now dosed without complications
- Generally well-tolerated with a favorable safety profile in five patients
- Serum beta-galactosidase enzyme activity increased in all patients at all timepoints between Day 7 and Month 6, representing an approximate doubling in enzyme activity after gene transfer
- At Month 6, enzyme activity restored to 23-57% (mean: 38%) of normal reference levels
- All five children demonstrated signs of clinical disease stability as assessed by Vineland-3 Growth Scale Value, Upright and Floor Mobility, and Clinical Global Impression (CGI) scales
- High-dose cohort initiated in November 2020; two patients now dosed without complications
Penafian
Maklumat dan penerbitan adalah tidak bertujuan, dan tidak membentuk, nasihat atau cadangan kewangan, pelaburan, dagangan atau jenis lain yang diberikan atau disahkan oleh TradingView. Baca lebih dalam Terma Penggunaan.
Penafian
Maklumat dan penerbitan adalah tidak bertujuan, dan tidak membentuk, nasihat atau cadangan kewangan, pelaburan, dagangan atau jenis lain yang diberikan atau disahkan oleh TradingView. Baca lebih dalam Terma Penggunaan.